


CONFERENCE OVERVIEW
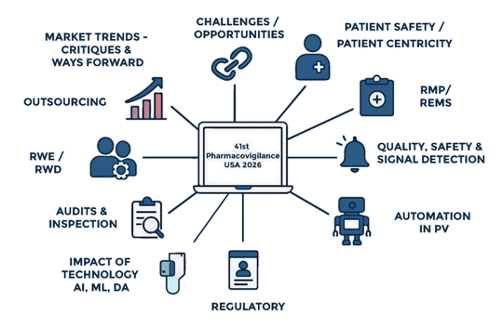
41st Pharmacovigilance USA 2026
Key Focus Areas
5 REASONS TO ATTEND THE EVENT
UNLOCK OPPORTUNITIES, INSIGHTS,
AND CONNECTIONS THAT MATTER
At Business, we bring together brightest minds, leaders, and trailblazers from across industries to explore latest trends, technologies, and strategies shaping the future.
Knowledge
Gain insights from experts, researchers, and professionals on the latest pharma advancements.Knowledge Gain insights from experts, researchers, and professionals on the latest pharma advancements.
An Agenda Made For You
High quality content that addresses your biggest challenges and uncovering hidden opportunities.Explore the latest trends, innovations, and breakthroughs driving the future of the pharmaceutical industry.
Cutting-Edge Insights
Explore the latest trends, innovations, and breakthroughs driving the future of the pharmaceutical industry.
Networking
Connect with peers, industry leaders, and collaborators to exchange ideas and explore new opportunities.
Practical Takeaways
Walk away with practical strategies, case studies, and proven best practices to apply in real-time.
Trusted by Industry Leaders Worldwide
events speakers
OUR EVENT SCHEDULE
09:00 – Chairperson opening remarks
Registration & Refreshments
Kick off your conference experience by checking in at our registration desk. Collect your event materials and enjoy a selection of refreshments. It’s the perfect time to mingle with fellow attendees and set the tone for an engaging and informative event

09:00 – Chairperson opening remarks

BEN LOCWIN
VP Board of Directors
SOPPhC (VP, Reliant Life Sciences)
09:10 – AI Agents Transforming Safety, Reporting, and
Operations
DATA TO DECISIONS
- Medical AI Agent for Adverse Event Assessment
- From Data to Draft: A Proof-of-Concept Agent for AI – Assisted Aggregate Report Writing
- An AI Agent for Intelligent Navigation of Company Procedures

PRIYANKA CHHIKARA
Head of Risk Management, Aggregate Reports & Data
Science, CSL Behring

VINEET SINGH
Head of PV Data Science and Analytics
CSL Behring
09:40 – Real World Data to Real World Evidence
RWD / RWE
- Importance of real-world data for answering regulatory questions
- Benefits and challenges of evidence derived from real world sources
- Real World Evidence: How Does its Use challenge quality and compliance programs?
- Obtaining patient-centric using RWE
- What are complications that will rise by inserting new data source? Breakup in data?
- Identify the challenges involved in incorporating real world evidence into a structured benefit-risk assessment.

JULIE BARBERIO
Director, Pharmacoepidemiology
Sanofi
10:10 – Keynote Panel Discussion: Reimagining Global Pharmacovigilance: From Today’s Challenges to Tomorrow’s Solutions
CHALLENGES & OPPORTUNITIES
Moderator:

MARIETTE BOERSTOEL-STREEFLAND
SVP, Worldwide Patient Safety Officer
Bristol-Myers Squibb
Panelists:

PHILISHA MACK
Sr. Director of Drug Safety, Medical Affairs
Azurity Pharmaceuticals

NICOLE SCHUMACHER-CROW
Continuous Improvement Center of Excellence Head –
Senior Director
Pfizer

MILIND SARDESA
Executive Director, Safety Surveillance, Global Patient
Safety,
Dicerna Pharmaceuticals

VASUDEV BHUPATHI
Director – Pharmacovigilance
Moderna

NAMITA JOSHI
Regional PV Head, North America
Sanofi
11:00 – Morning Networking Coffee/Tea & Discussion
11:30 – Gene therapy long-term safety management
PV IN GENE THERAPHY

MALLIKA SINGH
Associate Director, Pharmacovigilance
Genetix Biotherapeutics
12:00 – Solution Provider Presentation
For sponsorship opportunities please contact
info.uk@virtueinsight.com
12:30 – Networking luncheon
13:30 – Panel Discussion – Transforming Pharmacovigilance Through Technology: AI, Automation, and the Future of Drug Safety. Opportunities, Challenges & Lessons Learned
IMPACT OF TECHNOLOGY
Moderator:

HEATHER LEIGH FLANNERY
CEO & Co-Founder
AI MINDSystems Foundation
Panelists:

ALTON SARTOR
Medical Director, Patient Safety Physician Digital &
Devices
AstraZeneca

VIKALP KHARE
Director, Head of US GPV Safety Data Management
Otsuka Pharmaceutical

MICHAEL GLASER
Technology Innovation Director
GSK

PRIYANKA CHHIKARA
Head of Risk Management, Aggregate Reports & Data
Science
CSL Behring
14:20 – From Compliance to Excellence: The future of
Patient Safety
COMPLIANCE
- Patient Safety, where are we and what has changed lately?
- Fundamentals remain unchanged
- Shift the focus
- Implementation strategy
- Conclusion and take aways

JULIE BARBERIO
Director, Pharmacoepidemiology
Sanofi
14:50 – Gene therapy, long-term follow-up, and Inspection readiness
GENE THERAPY / INSPECTION
- Long-term follow-up requirements for Gene therapy
- How to plan inspection readiness activities for Gene therapy.
- Risk Management in Gene Therapy

KAPIL BHUTADA
Head of PV Safety Operation & Compliance
Asklepios BioPharmaceutical
15:20 – Afternoon Networking Coffee/Tea & Discussion
15:50 – The round table series
COLLABRATIVE BREAKOUT SESSIONS
Chairperson for all tables

BEN LOCWIN
VP Board of Directors
SOPPhC (VP, Reliant Life Sciences)

JYRKI SYVAERI
Corporate Director
Boehringer Ingelheim

JYRKI SYVAERI
Corporate Director
Boehringer Ingelheim
events sponsors
We wouldn’t be able to host our conferences without help from these amazing companies. A huge thanks to all our sponsors and partners!
Platinum
Pfizer

Amgen
Gold
Pfizer
Takeda
Takeda
Pullman Brussels Centre
Midi, Brussels, Belgium
Belgium
+32 2 528 98 00
Get Involved
Speaking Opportunities
Sponsorship &
Delegate Enquiry
EARLY BIRD PRICE - INDIVIDUAL BOOKING
- Entry to the Conference
- Motivating keynote sessions, dynamic panel discussions, engaging Q&As, and more with key industry leaders
- Valuable in-person networking and idea exchange with fellow professionals
- Innovative insights and cutting edge solutions from top-tier providers
- Full access to our networking platform
- Receive Certificate of Attendance
- Access to PDF presentations* via our website pending speaker permissions
- Prices inclusive of 21% VAT
EARLY BIRD PRICE - INDIVIDUAL BOOKING
- Entry to the Conference
- Motivating keynote sessions, dynamic panel discussions, engaging Q&As, and more with key industry leaders
- Valuable in-person networking and idea exchange with fellow professionals
- Innovative insights and cutting edge solutions from top-tier providers
- Full access to our networking platform
- Receive Certificate of Attendance
- Access to PDF presentations* via our website pending speaker permissions
- Prices inclusive of 21% VAT
We strive to produce high quality conferences which include the latest topics which are delivered by world class leaders of the industry.
UK
Block 3, 86 Coombe Road
Croydon CR0 5RA
London, UK
+44 20 3509 3779
info.uk@virtueinsight.com
Asia
Plot No – 07 – 2nd Floor
Ekambaram Industrial Estate
Alapakkam, Porur Chennai,
India – 600 116
+91 44421 08101
info.uk@virtueinsight.com
Quick Links
HOME
ABOUT US
EVENTS
EVENT SUPPORT
FAQ
CONTACT US
PRIVACY POLICY
TERMS & CONDITIONS
STAY INFORMED
Subscribe now to receive updates on our events






































